Abstract

Background: Pts with higher-risk MDS and AML need treatment options that provide durable responses with sustained clinical benefit and favorable safety/tolerability. Sabatolimab is a novel immuno-myeloid therapy targeting TIM-3, an immune regulator expressed on immune cells and myeloid leukemic progenitors but not on normal hematopoietic stem cells. Sabatolimab + HMA has been shown to deliver promising durable responses in a phase (Ph) Ib study in pts with vHR/HR-MDS or newly diagnosed (ND) AML (Wei et al. EHA 2021; NCT03066648). We report updated data from this study, including new analyses evaluating a potential relationship between immune-mediated effects of sabatolimab and response. Final results with additional follow-up will be reported at the time of presentation.
Methods: Eligibility criteria and design of this multicenter, open-label study have been previously reported (Wei et al. EHA 2021). The primary objective was to evaluate safety and tolerability. Preliminary efficacy, a key secondary objective, was assessed with overall response rate (ORR), duration of response (DOR), and progression-free survival (PFS) endpoints. Safety and preliminary efficacy are reported for sabatolimab + HMA in pts with vHR/HR-MDS (per IPSS-R) or ND-AML. The proportion of pts who had a possible immune-mediated adverse event (imAE) was evaluated among pts who achieved remission (CR + PR + CRi/mCR) compared with pts that have not achieved remission. The outcomes of pts who received hematopoietic stem cell transplantation (HSCT) after coming off the study were assessed independent of the study by investigators.
Results: As of the 15 Jun 2021 data cutoff, 53 pts with vHR/HR-MDS and 48 with ND-AML were treated with sabatolimab + HMA. The combination was safe and well tolerated, with the most common (≥15% in either vHR/HR-MDS or ND-AML) gr ≥3 AEs similar to HMA alone, consisting of thrombocytopenia (43.4%, 45.8%), neutropenia (47.2%, 50.0%), anemia (28.3%, 33.3%), and febrile neutropenia (35.8%, 29.2%), respectively. No pt with vHR/HR-MDS and only 3 with ND-AML discontinued treatment due to an AE regardless of relationship to treatment. In vHR/HR-MDS, 24.5% of pts had improvement allowing them to undergo HSCT. While on study, 6 pts with vHR/HR-MDS and 10 with ND-AML had possible imAEs regardless of relationship to study treatment. Few pts had clinically significant possible imAEs, with no gr ≥3 possible imAEs in pts with vHR/HR-MDS. In ND-AML, 5 pts had gr 3 and none had gr 4/5 possible imAEs.
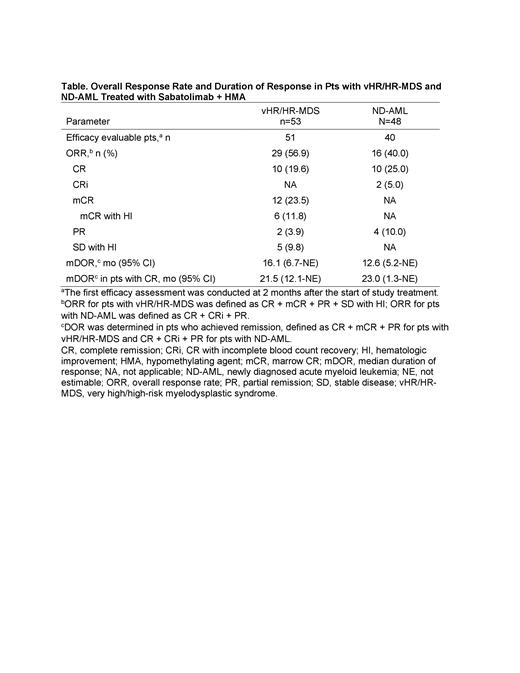
Among 51 pts with vHR/HR-MDS evaluable for response, ORR was 56.9%, with a median DOR (mDOR) of 16.1 mo (Table). The mDOR in pts with CR was 21.5 mo (95% CI, 12.1-NE). Estimated 12-mo PFS rate was 51.9% (95% CI, 30.6%-69.6%). In 40 evaluable pts with ND-AML, ORR was 40.0% and mDOR was 12.6 mo. The mDOR in pts with CR was 23.0 mo (95% CI, 1.3-NE). Estimated 12-mo PFS rate was 27.9% (95% CI, 14.9%-42.5%). Durable responses were also observed in pts with adverse-risk mutations, including TP53 mutations in pts with vHR/HR-MDS (ORR: 71.4% [10/14]; mDOR 21.5 mo [95%CI, 6.7-NE]) and at least 1 ELN adverse-risk mutation (TP53/RUNX1/ASXL1) in pts with ND-AML (ORR: 53.8% [7/13]; mDOR 12.6 mo [95%CI, 1.3-NE]).
Although the majority (75%) of pts who went into remission did not experience an imAE, pts in the vHR/HR-MDS cohort who achieved remission more often had a possible imAE (6/24 [25%]) than pts without remission (n=27), none of whom had an imAE. Notably, all 6 pts with vHR/HR-MDS who had an imAE achieved remission. Among pts with ND-AML, the frequency of possible imAEs was similar regardless of remission status. Of the subset of pts who proceeded to HSCT, post-HSCT outcomes in pts with vHR/HR-MDS treated with sabatolimab + HMA were generally favorable without excess toxicities related to graft-versus-host disease.
Conclusions: Sabatolimab + HMA was safe and well tolerated and demonstrated durable clinical responses in pts with vHR/HR-MDS and ND-AML. Responses were also durable in pts with adverse-risk mutations. The observed relationship between response and possible imAEs in vHR/HR-MDS would need further confirmation in ongoing studies, but suggests that an immunomodulatory mechanism of sabatolimab may be contributing to clinical responses. The STIMULUS clinical trial program is evaluating sabatolimab-based combination therapy in multiple Ph II and III studies in MDS and AML.
Co-senior authors Uma Borate and Andrew H. Wei contributed equally.
Brunner: Celgene, Forty Seven Inc, Jazz: Other: Advisory Board; Novartis, Celgene, Takeda, AstraZeneca: Research Funding. Esteve: Bristol Myers Squibb/Celgene: Consultancy; Abbvie: Consultancy; Novartis: Consultancy, Research Funding; Astellas: Consultancy; Jazz: Consultancy; Pfizer: Consultancy; Novartis: Research Funding. Knapper: Jazz Pharmaceuticals: Consultancy, Speakers Bureau; Astellas: Consultancy, Speakers Bureau; Novartis: Consultancy, Research Funding, Speakers Bureau; Pfizer: Consultancy, Speakers Bureau. Traer: Schrodinger: Research Funding; ImmunoGen: Membership on an entity's Board of Directors or advisory committees; Astellas: Consultancy, Membership on an entity's Board of Directors or advisory committees; Servier/Agios: Membership on an entity's Board of Directors or advisory committees; Incyte: Research Funding; Abbvie: Consultancy, Membership on an entity's Board of Directors or advisory committees; Genentech: Membership on an entity's Board of Directors or advisory committees. Vey: Amgen: Honoraria; BMS: Honoraria; BIOKINESIS: Consultancy, Research Funding; NOVARTIS: Consultancy, Honoraria, Research Funding; SERVIER: Consultancy; JAZZ PHARMACEUTICALS: Honoraria; JANSSEN: Consultancy. Wermke: Novartis, Roche, Pfizer, BMS: Consultancy, Honoraria, Research Funding. Janssen: Uppsala County Council: Research Funding; Celgene: Membership on an entity's Board of Directors or advisory committees, Speakers Bureau; Bristol-Myers Squibb: Consultancy, Research Funding; Roche: Speakers Bureau; Avillion: Research Funding; Ellipses Pharma: Research Funding; Novartis: Consultancy, Research Funding; Pfizer: Consultancy; Incyte Biosciences Benelux BV: Research Funding, Speakers Bureau; Glycomimetics: Research Funding. Narayan: Novartis: Research Funding; Sanofi Genzyme: Other: Spouse employment & equity interest; Takeda: Other: Spouse employment & equity interest; Genentech: Other: Spouse employment & equity interest. Kontro: Jazz Pharmaceuticals: Membership on an entity's Board of Directors or advisory committees; Astellas: Consultancy, Membership on an entity's Board of Directors or advisory committees; AbbVie: Membership on an entity's Board of Directors or advisory committees, Research Funding. Ottmann: Celgene/BMS: Honoraria, Research Funding; Fusion: Honoraria; Novartis: Honoraria; Amgen: Honoraria, Research Funding; Incyte: Honoraria, Research Funding. Naidu: Novartis: Current Employment. Pelletier: Novartis: Current Employment. Han: Novartis: Current Employment, Current equity holder in publicly-traded company. Lewandowski: Novartis Institutes: Current Employment. Zhang: Novartis Institutes for BioMedical Research: Current Employment. Mohammed: Novartis: Current Employment. Rinne: Novartis: Current Employment; Qiagen: Consultancy. Borate: Daiichi-Sankyo: Membership on an entity's Board of Directors or advisory committees; Pfizer: Membership on an entity's Board of Directors or advisory committees, Research Funding; Genentech: Membership on an entity's Board of Directors or advisory committees, Other: Advisory Board; AbbVie: Membership on an entity's Board of Directors or advisory committees, Research Funding; Novartis: Membership on an entity's Board of Directors or advisory committees, Research Funding; Blueprint Medicine: Membership on an entity's Board of Directors or advisory committees; Takeda: Membership on an entity's Board of Directors or advisory committees; Jazz Pharma: Research Funding; Astellas: Membership on an entity's Board of Directors or advisory committees; incyte: Membership on an entity's Board of Directors or advisory committees, Research Funding; Rampal: Membership on an entity's Board of Directors or advisory committees; Galecto, Inc.: Consultancy; Promedior: Consultancy. Wei: Novartis, Janssen, Amgen, Roche, Pfizer, Abbvie, Servier, BMS, Macrogenics, Agios, Gilead: Membership on an entity's Board of Directors or advisory committees; Novartis, Celgene, AbbVie, Servier, AstraZeneca, and Amgen: Research Funding; Astellas: Honoraria.
Sabatolimab is a novel immuno-myeloid therapy targeting TIM-3 and is under investigation for the treatment of patients with myeloid malignancies
Author notes
 This icon denotes a clinically relevant abstract
This icon denotes a clinically relevant abstract

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal